However, Albert Einstein did not agree with this idea, and they debated their views at conferences in the late 1920s. Heisenberg and Bohr studied the Complementarity Principle, which is the idea that atoms must be studied both as small particles and as waves of energy.
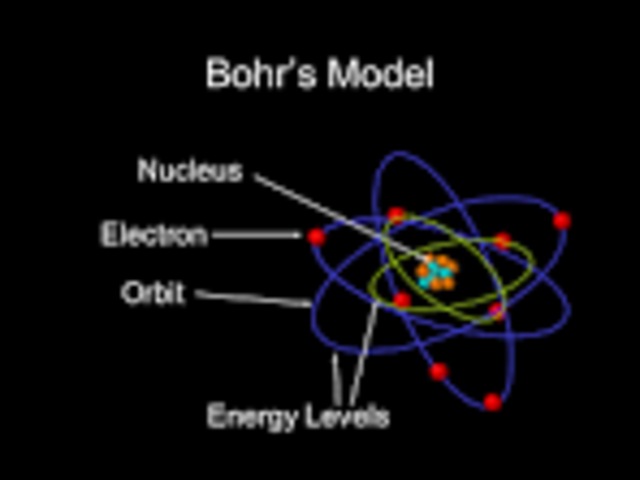
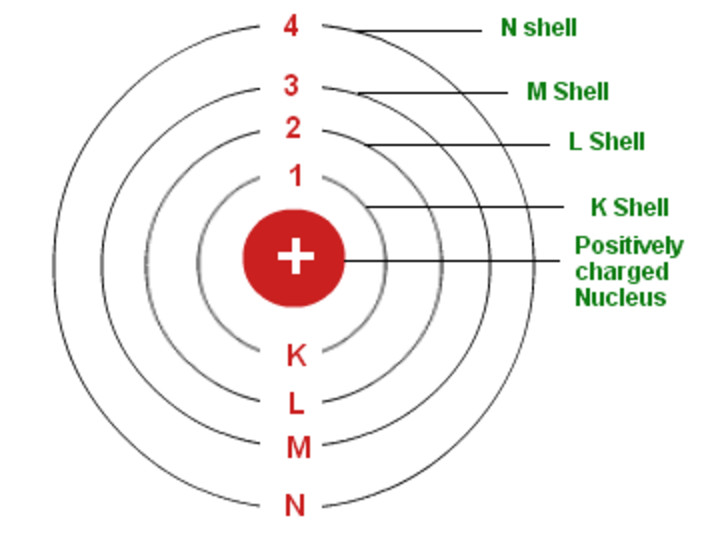
Werner Heisenberg became Bohr’s assistant at the University of Copenhagen. Shortly after, scientists were able to bring Bohr’s research on electron theory and the new atomic model to their research in a new field of physics called quantum mechanics. Alfred Nobel Scientific Discovery: Quantum Mechanics The effectiveness of the Bohr Atomic Model earned Niels Bohr the 1922 Nobel Prize. This work helped show that his model was accurate, and it was used in the process of discovering a new element, called Hafnium. Using his atomic model, Bohr added electrons to the periodic table. This later became known as the Bohr Atomic Model.Īfter becoming a professor of physics at Copenhagen, he established the Institute for Theoretical Physics, which opened in 1921.įor the next phase of his research, Bohr wanted to study the electron theory as it related to the periodic table of elements. They showed that tiny particles called electrons were negative and orbited around the center of the atom.īohr contributed to this research by writing journal articles for The Philosophical magazine and explaining that electrons specifically orbit in preset patterns.Īdditionally, he described how the orbiting electrons might release energy when they drop down to a lower orbit, and called this energy electromagnetic radiation. There, he focused on quantum physics, a new type of small particle physics at the time. Related: Molecule Quiz Scientific Discovery: The Atomic ModelĪfter earning his doctoral degree, Bohr continued his research at the University of Manchester, studying under Ernest Rutherford. His research focus during this time was the electron theory, which states that all matter is comprised of molecules, which in turn consist of protons, neutrons and electrons.

He followed in his father’s footsteps and became a University of Copenhagen student at age 18, and earned a PhD in physics 8 years later. He knew that he would have to do very well to understand physics if he wanted to do his own research. He worked very hard in school as a child so that he could go to the same university as his father. He was the middle child of a University of Copenhagen physiology professor, but he knew that he wanted to be a physicist (scientist who studies physics) early on in his life.

